
Washington, D.C., USA- January 13, 2020: FDA Sign at its headquarters in Washington DC. The Food and Drug Administration (FDA or USFDA) is a federal agency of the USA.
‘Good Brain Tonic’ Pulled from Shelves Over Potential Botulism Contamination
A New York-based beverage company is recalling all bottles of its “Good Brain Tonic” after state and university testing detected a potential risk of botulism, a rare but potentially fatal form of food poisoning.

Liquid Blenz Corp of Rockville Center, New York, announced the voluntary recall on April 6, 2026, according to a company statement published by the U.S. Food and Drug Administration. The recall covers all production codes of Good Brain Tonic sold in 16-ounce and 32-ounce amber glass bottles with plastic caps.
The product was distributed nationwide through retail stores and direct-to-consumer internet sales. The UPC code for the 16-ounce bottle is 860010984468, and the 32-ounce bottle is 860010984475.
According to the company announcement, the potential for botulism contamination was identified through analysis conducted by the Cornell Food Venture Center and subsequent field testing by food safety inspectors from the New York State Department of Agriculture and Markets.
Botulism is a serious illness caused by a toxin that attacks the body’s nerves. Symptoms can include general weakness, dizziness, double vision, and difficulty speaking or swallowing. As the illness progresses, patients may experience trouble breathing, muscle weakness, abdominal distension, and constipation. Health officials advise anyone experiencing these symptoms after consuming the product to seek immediate medical attention.
No illnesses have been reported to date in connection with the recalled tonic, the company stated.
Liquid Blenz Corp is directing consumers who purchased the product to return it to the place of purchase for a full refund. Customers with questions can contact the company directly at 1-516-608-8826.
Popular Amazon Nail Polish Remover Recalled After FDA Tests Find Cancer-Linked Chemicals in Formula

A gel nail polish remover sold widely on Amazon is being recalled after federal regulators confirmed the product contains methylene chloride and chloroform—two hazardous and strictly prohibited substances that cannot legally be used in cosmetic formulations in the United States.
The recall involves approximately 4,000 units of Morovan brand Gel Nail Polish Remover, distributed in 15-milliliter square turquoise bottles with white caps and matching green packaging boxes. The product was sold exclusively through Amazon from August 2025 through January 2026, according to a company announcement published by the U.S. Food and Drug Administration.
Testing detected the presence of methylene chloride, a solvent that has been shown to cause cancer in animal studies and is considered likely harmful to human health. Under FDA regulations, methylene chloride is a banned ingredient prohibited at any concentration in cosmetic products. The presence of chloroform raises similar health concerns.
The recalled items bear a production date of February 7, 2024, and an expiration date of February 7, 2027. At the time of this reporting, no incidents or injuries have been reported to date in connection with the recalled remover.
Consumers who purchased the product are instructed to stop using it immediately. The company is directing customers to clearly write the word “Recalled” on both the product bottle and its outer packaging, then dispose of the entire item in regular household waste according to local disposal guidelines. After disposal, customers must submit a photograph showing the labeled and discarded product to the company at customer-service@morovan.com to verify compliance and obtain a refund.
The product was manufactured in China and distributed by HONGKONG YIIXIN TRADING LIMITED, also identified as Nails Studio. The recall notice was published by the FDA on April 13, 2026, following a company announcement dated April 9.
Blueroot Health Widens Recall of Vital Nutrients Allergy Supplement Over Undeclared Egg, Hazelnut, and Soy Contamination
A Connecticut supplement manufacturer has significantly expanded a recall of its popular allergy support capsules, warning that the product may contain three undeclared allergens that pose a serious or life-threatening risk to sensitive individuals.
Blueroot Health of Middletown, Connecticut, announced on April 10, 2026, that it is broadening a voluntary recall of Vital Nutrients Aller-C dietary supplements to include all product manufactured from lot number 25E04. The expansion follows an initial recall issued on March 27, 2026, which was limited to sub-lots 25E04-A and 25E04-B.
The recalled capsules may contain undeclared egg, hazelnut, and soy. People who have allergies or severe sensitivity to any of these ingredients run the risk of a serious allergic reaction, including anaphylaxis, if they consume the product.
According to the company’s announcement, the impacted lots were distributed nationwide through VitalNutrients.co and other online retailers between September 2025 and March 2026. The supplement is sold in white plastic bottles containing either 100 or 200 capsules. Affected bottles bear the lot numbers 25E04, 25E04-A, or 25E04-B, along with an expiration date of 05/27 printed on the side of the bottle.
The presence of the undeclared allergens was discovered through routine internal testing and promptly reported to the U.S. Food and Drug Administration. No illnesses have been reported to date in connection with the recalled product.
Consumers who purchased Vital Nutrients Aller-C from the affected lots are instructed to immediately stop using the product and return it to Blueroot Health for a replacement. Customers with questions may contact the company at (888) 328-9992, Monday through Friday from 8:00 a.m. to 7:00 p.m. Eastern Time, or via email at support@vitalnutrients.co.
| Product | Count | UPC | Lot Numbers | Expiration Date |
|---|---|---|---|---|
| Vital Nutrients Aller-C | 200 | 693465524213, 693465000090 | 25E04-B | 05/27 |
| Vital Nutrients Aller-C | 100 | 693465524114, 693465000083 | 25E04, 25E04-A, 25E04-B | 05/27 |




Nationwide Recall Issued for Antiseptic Wound Gels Due to Bacterial Contamination, Risk to Immunocompromised Patients
A California pharmaceutical company is recalling thousands of tubes and bottles of antiseptic wound care gel distributed to doctors’ offices nationwide after internal testing detected contamination with an environmental bacterium that could cause severe infections in vulnerable patients.
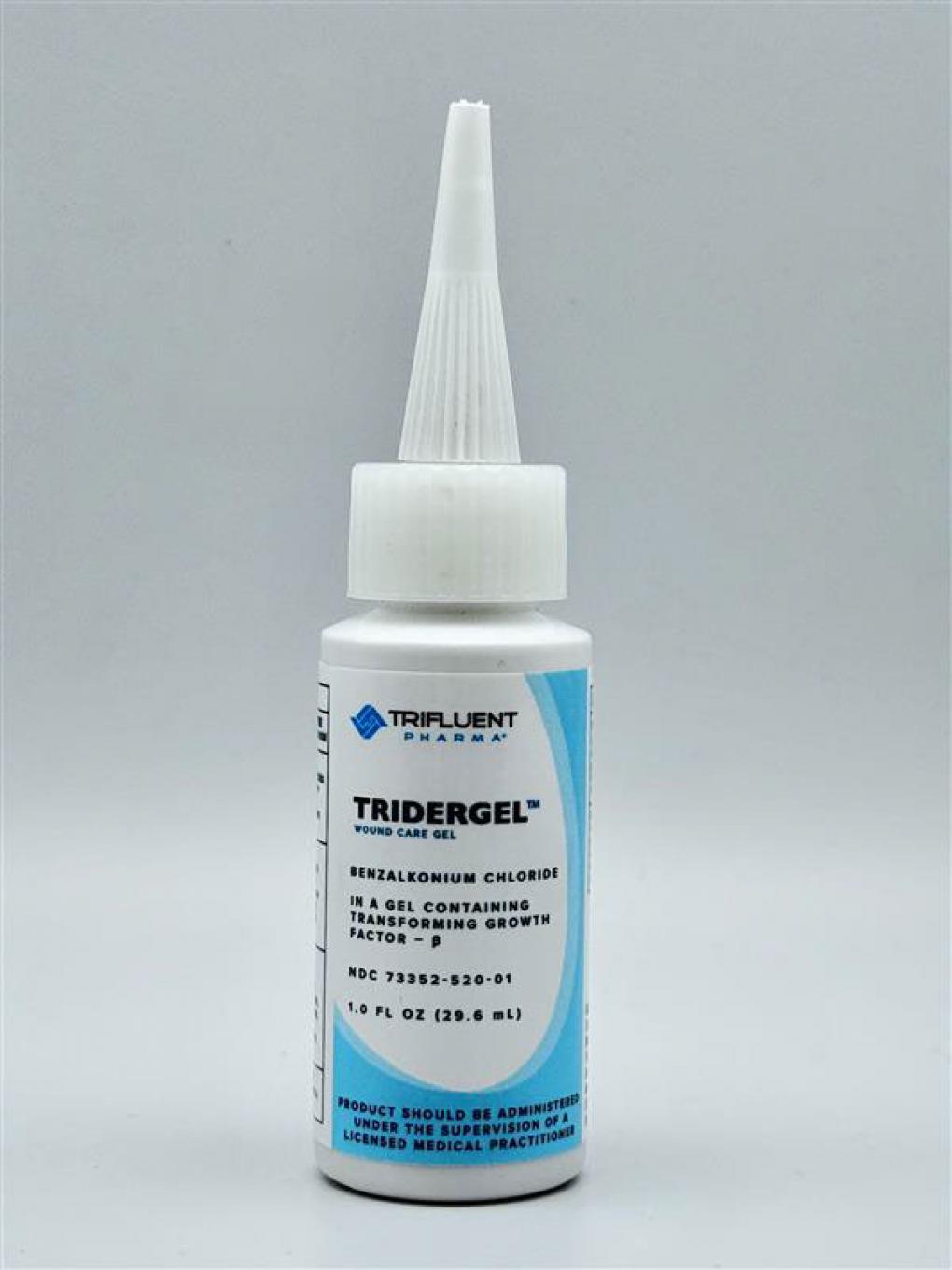
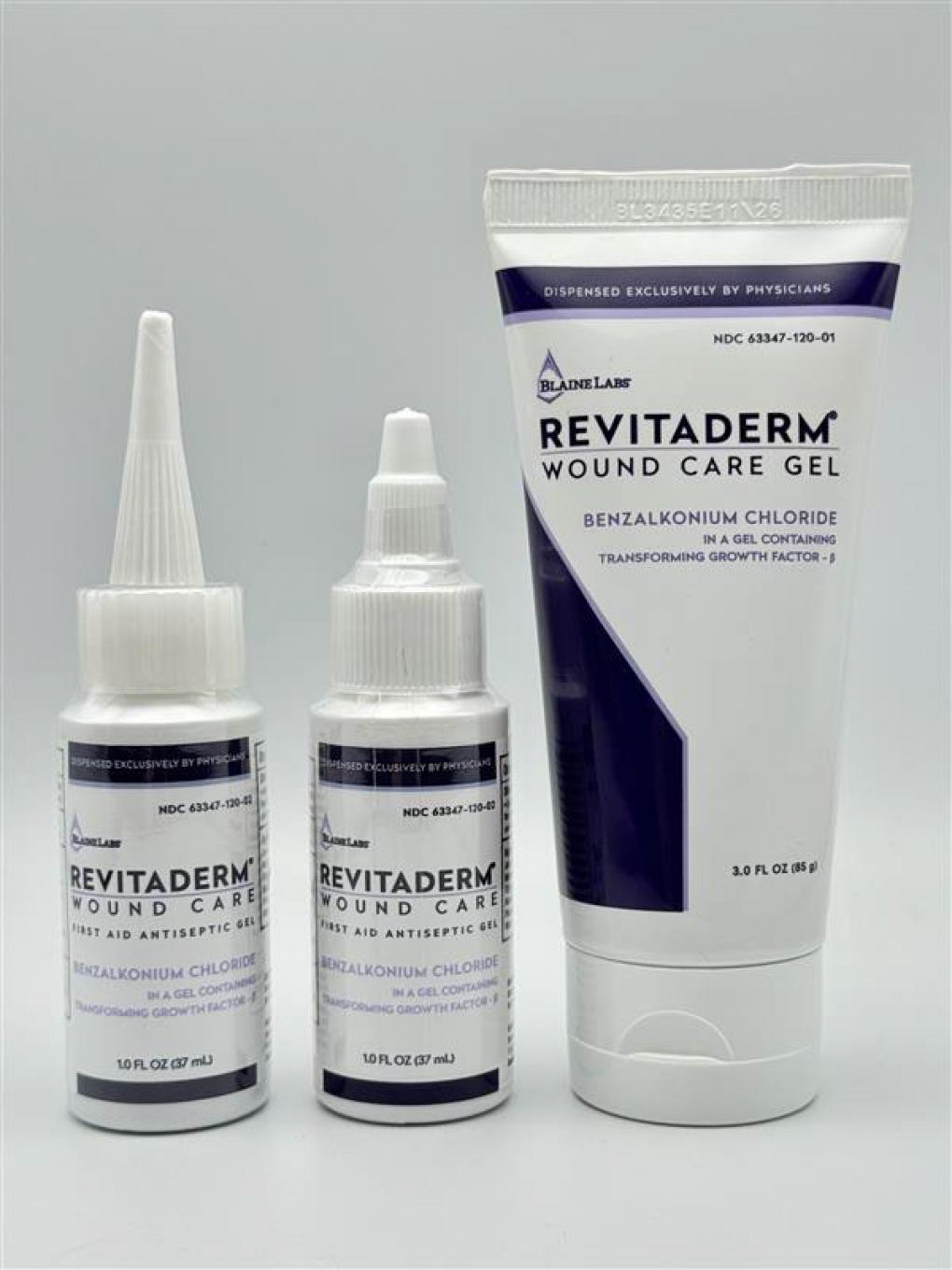
Blaine Labs, Inc. of Santa Fe Springs announced the voluntary recall on April 8, 2026, affecting three lot numbers of Revitaderm and Tridergel brand wound care gels containing 0.1% benzalkonium chloride. The affected products were distributed exclusively to healthcare providers and were not sold at retail locations or online.
Laboratory analysis identified the presence of Lysinibacillus fusiformis, a spore-forming environmental organism. While the bacterium is generally considered to have low pathogenicity in healthy individuals, federal health officials warn that it poses a heightened risk to patients with open wounds, compromised skin barriers, or weakened immune systems.

According to the company’s recall notice, immunocompromised individuals—including the elderly, diabetics, and young children—face an increased risk of localized skin infections, delayed wound healing, and in severe cases, life-threatening complications such as endocarditis or central nervous system infection.
The recall encompasses Revitaderm Wound Care Gel in 1-ounce bottles and 3-ounce tubes, identifiable by purple trim on the packaging, and Tridergel Wound Care in 1-ounce bottles with light blue trim. Affected lot numbers include BL3608 with an expiration date of July 1, 2028; BL3435 expiring November 6, 2026; and BL3525 expiring August 7, 2027. Lot numbers and expiration dates are printed on the bottom of bottles or imprinted on the crimp of tubes.
The products were distributed to physician clinics across the United States. Blaine Labs reported that it has not received any reports of adverse events related to the recalled gels.
Healthcare providers are instructed to immediately stop using and distributing the affected products, segregate remaining inventory, and contact Blaine Labs to arrange for return and replacement. The company is notifying physician offices by mail and telephone.
Consumers who may have received the product from a healthcare provider can contact Blaine Labs directly at (800) 307-8818, Monday through Friday from 7:30 a.m. to 4:00 p.m. Pacific Time, or via email at Contact@blainelabs.com. Adverse reactions may be reported to the FDA’s MedWatch program.
The recall is being conducted with the knowledge of the U.S. Food and Drug Administration.

